Sodium chloride is an ionic compound composed of sodium cations, Na +, and chloride anions, Cl −, combined in a 1:1 ratio. Keep in mind, however, that the formula for an ionic compound does not represent the composition of a discrete molecule, so it may not correctly be referred to as the “molecular mass.”Īs an example, consider sodium chloride, NaCl, the chemical name for common table salt. The formula mass for an ionic compound is calculated in the same way as the formula mass for covalent compounds: by summing the average atomic masses of all the atoms in the compound’s formula. Ionic compounds are composed of discrete cations and anions combined in ratios to yield electrically neutral bulk matter. A black sphere, attached to two red spheres and a white sphere is attached to the black sphere on the top right of the six-sided ring. Attached to the first black sphere of that row is another red sphere. Attached to the last black sphere of that row are two more white spheres. Attached to the farthest right black sphere is a red sphere, connected to two more black spheres, all in a row. Attached to each of the four black spheres is one smaller white sphere. Six black spheres are located in a six-sided ring and connected by alternating double and single black bonds. The merged cell under the first five columns reads “Molecular mass.” To the left of the table is a diagram of a molecule. The fifth column contains the symbol “=” in each cell except for the last, merged cell. The fourth column contains the numbers “12.01,” “1.008,” and “16.00” as well as the merged cell. The third column contains the multiplication symbol in each cell except for the last, merged cell. The second column contains the numbers “9,” “8,” and “4” as well as the merged, cell. The merged cell runs the length of the first five columns. The header row reads: “Element,” “Quantity,” a blank space, “Average atomic mass (a m u),” a blank space, and “Subtotal (a m u).” The first column contains the symbols “C,” “H,” “O,” and a merged cell. The table is made up of six columns and five rows. The model shows the molecular structure of aspirin, C 9H 8O 4. The accompanying table lists some useful hydrates.\): The average mass of an aspirin molecule is 180.15 amu. Aluminum chloride hexahydrate is an active ingredient in antiperspirants.   :max_bytes(150000):strip_icc()/GettyImages-175532236-c614b233b7e84d5487cad8b280f365a4.jpg)
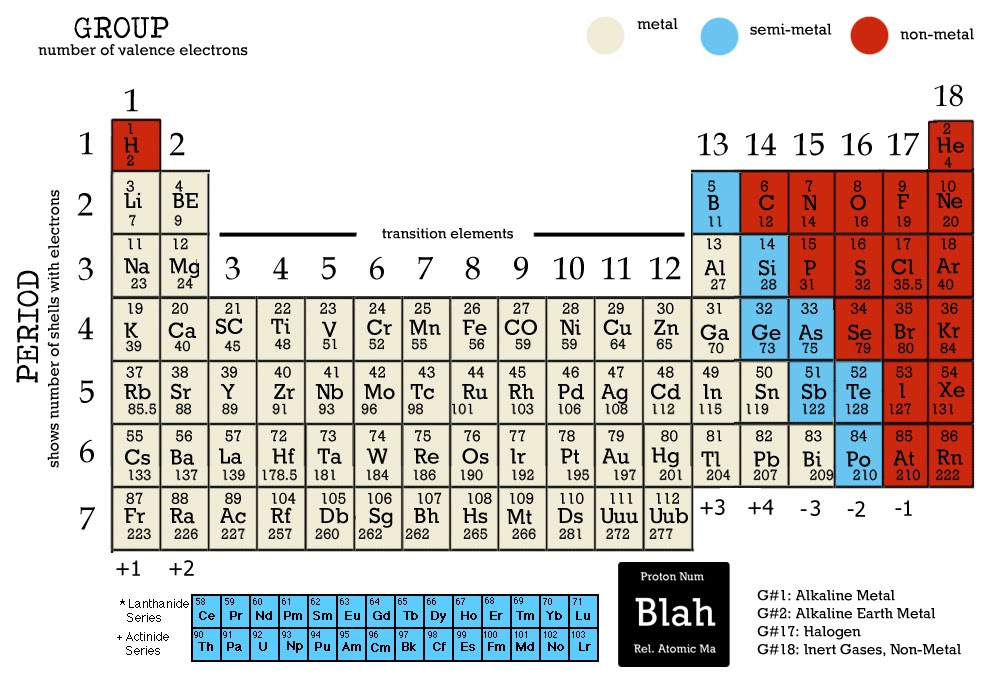
Epsom salt (MgSO 4♷H 2O) is used as a bathing salt and a laxative. The periodic table is arguably the best tool in all of science no other branch of science can. The periodic table lists all the known chemical elements, arranged by atomic number (that is, the number of protons in the nucleus). In this chapter, we present some data on the chemical elements. ½H 2O), known as plaster of Paris, is used to make casts for broken bones. Appendix: Periodic Table of the Elements.Hydrates have various uses in the health industry. The name of this compound is copper(II) sulfate pentahydrate, with the penta- prefix indicating the presence of five water units per formula unit of copper(II) sulfate. The hydrate of copper(II) sulfate has five water units associated with each formula unit, so it is written as CuSO 4♵H 2O. The two chemical formulas are separated by a vertically centered dot. To write the chemical formula of a hydrate, write the number of water units per formula unit of compound after its chemical formula. Hydrates are solids, not liquids or solutions, despite the water they contain. These compounds, called hydrates, have a characteristic number of water units associated with each formula unit of the compound. Some ionic compounds have water (H 2O) incorporated within their formula unit.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
